Distillation
What it is about
Distillation is a process that separates a volatile liquid from less-volatile liquid. First there has to be some source of heating, and containment of those vapors, and then, there needs to be a cooling system that cools the more volatile liquid into a separate container.
The equipment
While traditional chemistry set ups [1] can look very specific, as long as it maintains the above mentioned conditions, the configuration can be quite simple.
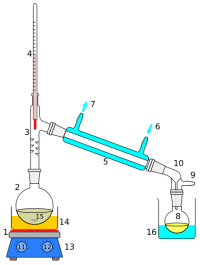
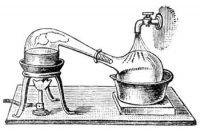
Our set up in Bangalore were pieces all bought from Shivajinagar in the local markets.
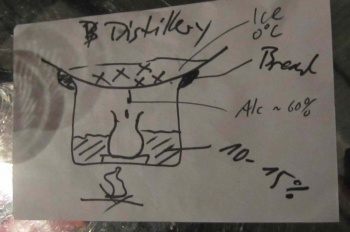



The Process
First, you have to have something to distill. We decided to distill a local wine. Here, ethanol (boiling point at 78.1C at 1 atmosphere) should evaporate first, separating from the water (boiling point at 100C).
Then we nested a cup inside the big pot, and put a rounded plate as a lid. The plate is best if the bottom is rounded, so the distilled alcohol can drip into the cup as in the sketch above.
We then need to seal the plate and the pot very well. We used bread mashed up with water to make the seal here.


If the seal is not complete, the more volatile liquid will escape, and you cannot collect it.
Now we are ready to distill - fire up the heat:
We did not have any ice to keep the top bowl cool, so Arun had to keep changing the water.
Voila! The original wine was in the bigger container, and the distilled spirit is in the cup in the middle (here, taken out). We drank it, but it is without all of the wine flavor...a bit difficult to enjoy.



