Woven actinometer for quantum
I have been noticed that there are many potassium ferrioxalate crystals formed and "left over" from making cyanotype sensitizer for my laser dye project, the crystal is relatively easy to make and can be molded so I am thinking to build a crystal installation in large size. then I found it's the chemical for the actinometer. However I am using "new cyanotype" invented by Mike Ware, this crystal are not found in the traditional cyanotype formula. By mixing the two basic solutions potassium ferricyanide and ferric ammonium oxalate with heat, the crystal is formed after placing the mixed solutions for 2 hours to 48 hours, the duration length is relevant to the crystal physical shape, usually the longer the bigger crystal. Below I tried to "grow" the potassium ferrioxalate crystal on an aluminum plate and silk fibers.
what is actinometer?
then I am looking into this old technology actinometer.
Actinometers are instruments used to measure the heating power of radiation. They are used in meteorology to measure solar radiation as pyrheliometers.
These are parts from balloon-borne pyrheliometer #2 designed by Charles Greeley Abbot and built by his craftsman Andrew Kramer at the original Smithsonian Astrophysical Observatory in about 1913. Following designs that had been developed for meteorographs, the recording medium was sensitized photographic paper on a slowly rotating drum. The drum was encased in a light-tight chamber save for an entrance slit parallel to the diameter. A thermometer filled the slight so that the mercury level would be recorded by the rotating paper as a function of time, and of altitude, as recorder by a barometric needle whose shadow was also superimposed. Other clockdriven elements periodically exposed the bulb of the thermometer to direct skylight and to ambient air temperature. This instrument stands as a milestone in sophistication and ingenuity in early self-registering automata. The balloon-sondes were lofted from Catalina Island off the coast of California and some reached 25 kilometers altitude. Data from these devices led to greatly refined values of Langley's solar constant, and helped to establish the modern range. A complete unit (#3) is preserved at the National Museum of American History. These parts were retrieved from the stores of the original SAO workshops, housed in the Garber facility, in the mid-1980s. Picture taken at the National Air and Space Museum's Steven F. Udvar-Hazy Center in Chantilly, Virginia, USA.
An actinometer is a chemical system or physical device which determines the number of photons in a beam integrally or per unit time. This name is commonly applied to devices used in the ultraviolet and visible wavelength ranges. For example, solutions of iron(III) oxalate can be used as a chemical actinometer, while bolometers, thermopiles, and photodiodes are physical devices giving a reading that can be correlated to the number of photons detected.. The actinometer was invented by John Herschel in 1825; he introduced the term actinometer, the first of many uses of the prefix actin for scientific instruments, effects, and processes. The actinograph is a related device for estimating the actinic power of lighting for photography. Potassium ferrioxalate is commonly used, as it is simple to use and sensitive over a wide range of relevant wavelengths (254 nm to 500 nm). Other actinometers include malachite green leucocyanides, vanadium(V)–iron(III) oxalate and monochloroacetic acid, however all of these undergo dark reactions, that is, they react in the absence of light. This is undesirable since it will have to be corrected for. Organic actinometers like butyrophenone or piperylene are analysed by gas chromatography. Other actinometers are more specific in terms of the range of wavelengths at which quantum yields have been determined. Reinecke’s salt K reacts in the near-UV region although it is thermally unstable. Uranyl oxalate has been used historically but is very toxic and cumbersome to analyze.
Quantum Yield
Chemical actinometry involves measuring radiant flux via the yield from a chemical reaction. It requires a chemical with a known quantum yield and easily analyzed reaction products. The quantum yield (Φ) of a radiation-induced process is the number of times a specific event occurs per photon absorbed by the system. The "event" is typically a kind of chemical reaction. In optical spectroscopy, the quantum yield is the probability that a given quantum state is formed from the system initially prepared in some other quantum state. For example, a singlet to triplet transition quantum yield is the fraction of molecules that, after being photoexcited into a singlet state, cross over to the triplet state.
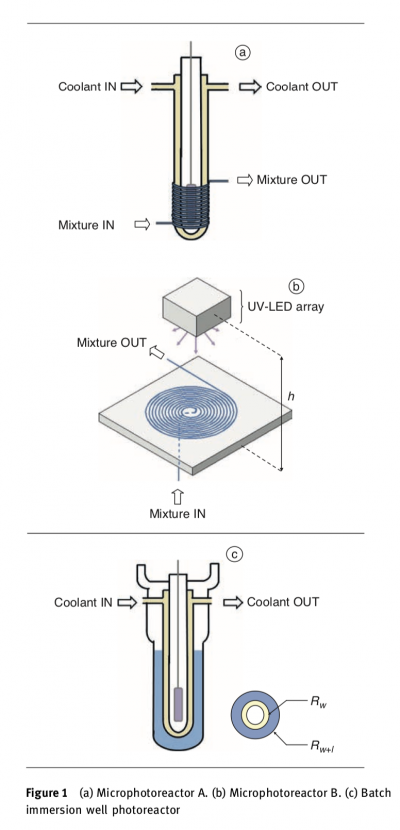
Then I found this device introduced in this paper: Accurate Measurement of the Photon Flux Received Inside Two Continuous Flow Microphotoreactors by Actinometry
The two continuous flow microphotoreacters reminds me some woven droplet tubes installation, for example here is the work of Victoria Manganiello, she weave these tubes and move the colored liquid in it by a programmed pump.