Difference between revisions of "GFP Protocol Jugaad"
| Line 203: | Line 203: | ||
. [[File:MG_7174.JPG]] | . [[File:MG_7174.JPG]] | ||
| − | [[File:MG_7028.JPG]] | + | [[File:MG_7163.JPG]] [[File:MG_7028.JPG]] |
| + | |||
| + | |||
| Line 209: | Line 211: | ||
'''Transformation:''' | '''Transformation:''' | ||
| − | ''' | + | |
| − | 16.''' Take a 2 vials of ligated sample and control DNA and 1 vial of only control DNA( positive control). | + | '''16.''' Take a 2 vials of ligated sample and control DNA and 1 vial of only control DNA( positive control). |
To this add 200 μl each of competent cells. Tap the | To this add 200 μl each of competent cells. Tap the | ||
vials gently and incubate on ice for 20 minutes. | vials gently and incubate on ice for 20 minutes. | ||
| Line 230: | Line 232: | ||
bacteria to recover and express the protein. | bacteria to recover and express the protein. | ||
| − | '''21.''' Pipette 200 μl each from the vials transformed with the | + | '''Note:''' While the heat shock treatment is going on, prepare the LB-Amp agar(200 μl in 200 ml of LB agar.) |
| + | |||
| + | [[File:MG_7064.JPG]] [[File:MG_7133.JPG]] | ||
| + | |||
| + | [[File:MG_7142.JPG]] [[File:MG_7143.JPG]] | ||
| + | |||
| + | [[File:MG_7174.JPG]] [[File:MG_7175.JPG]] | ||
| + | |||
| + | [[File:MG_7177.JPG]] [[File:MG_7174.JPG]] | ||
| + | |||
| + | [[File:MG_7185.JPG]] [[File:MG_7191.JPG]] | ||
| + | |||
| + | [[File:MG_7190.JPG]] [[File:MG_7186.JPG]] | ||
| + | |||
| + | .(incubate pic) | ||
| + | |||
| + | |||
| + | |||
| + | '''21.''' liquify the already made LB-amp agar and pour about 20 to 25 ml of i in each plate. | ||
| + | |||
| + | '''Note:''' Wait for agar to solidify again. | ||
| + | |||
| + | '''22.''' Pipette 200 μl each from the vials transformed with the | ||
ligated mix onto LB-Amp plates and spread thoroughly | ligated mix onto LB-Amp plates and spread thoroughly | ||
using spreader/pipette. | using spreader/pipette. | ||
| − | ''' | + | '''23.''' Pipette 200 μl of LB onto a LB -Amp plate and spread |
20 ìl of the cells transformed with control DNA. Label | 20 ìl of the cells transformed with control DNA. Label | ||
this as positive control plate. | this as positive control plate. | ||
| − | ''' | + | '''24.''' Plate 200 μl of non-transformed cells onto another |
LB-Amp plate to check for contamination. Label this | LB-Amp plate to check for contamination. Label this | ||
as non-transformed (negative control) plate. | as non-transformed (negative control) plate. | ||
| − | ''' | + | '''25.''' Keep one plate with only LB-Amp Agar and nothing else. Name this as double negative control. |
| + | |||
| + | '''26.''' Incubate the plates at 37°C, overnight. | ||
| + | |||
| + | |||
| + | [[File:MG_7204.JPG]] [[File:MG_7206.JPG]] | ||
| + | |||
| + | [[File:MG_7213.JPG]] [[File:MG_7221.JPG]] | ||
| + | |||
| + | [[File:MG_7225.JPG]] pippet out on plate | ||
| − | + | spreader incubate | |
| Line 253: | Line 286: | ||
'''Moment of Truth''' | '''Moment of Truth''' | ||
| − | ''' | + | '''27.''' Observe the plates under UV-light (312 nm). |
'''Note:''' If observed add 254 nm of less intensity of glow | '''Note:''' If observed add 254 nm of less intensity of glow | ||
could vary. | could vary. | ||
Revision as of 10:10, 4 September 2010
Materials
The list below provides information about the materials supplied in the kit.
The products should be stored as suggested. Use the kit within 6 months of arrival.
Materials……………………... Quantity………….store at
Ampicillin………………………..100 mg………….…..4°C
Host ………………………………1 vial……..………...4°C
Solution A………………………. 40 ml…………….….4°C
Control DNA……………………...10 μl….…………..-20°C
T4 DNA Ligase………………….…5 μl………………-20°C
10X Ligase Assay Buffer ….……10 μl……….……..-20°C
Insert………………………………20 μl………..…....-20°C
Vector DNA………………………10 μl ………….….-20°C
LB Broth……………………….....15 g ……………..…RT
Agar………………………………...5 g……..………... RT
1.5 ml vials……………………….25 Nos……….….... RT
Equipment :
-Centrifuge (preferably refrigerated)( DIY handheld centrifuge ).
-UV transilluminator (312 nm).
-Spectrophotometer.
Glassware :
-Capped centrifuge tubes.
-Conical flask.
-Petri plates.
-Test tubes.
Reagent :
-Distilled water.
Other Requirements:
-Crushed ice.
-Cuvette (of 1 cm pathlength).
-Micropipette, Tips.
-Thermometer.
-Water bath. ( DIY Water Bath )
-Incubator.(DIY Incubator )
-Sterilization Hood. ( DIY Sterlisation Hood )
Procedure:
Day 1:
Revival of Host
1. Break open the lyophilized vial, add 0.1 ml of LB broth.
2. Streak a loopful of the suspension onto LB plate & LB culture tubes. (in duplicates).
3. Incubate the plates at 37°C, overnight.
Ligation of Vector to Insert
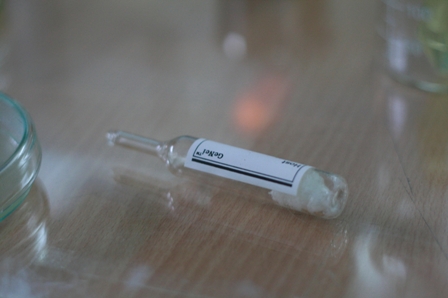
4. Thaw the ligase assay buffer, vector and insert DNA.
Note: Thaw the ligase assay buffer vial on ice, store at -20°C immediately after use.
5. Set up ligation reaction as follows:
-Water : 11 μl
-Vector DNA : 2 μl
-Insert DNA : 4 μl
-Ligase assay buffer : 2 μl
-T4 DNA ligase : 1 μl
Mix the contents by tapping gently and incubate at 16°C waterbath, overnight.
Note: Set up five ligation reactions simultaneously.
Day 2:
Preparation of Competent Cells
6. Incubate the culture tubes at 37°C. Grow until OD A600 reaches 0.3, this takes about 2-3 hours.
7. Chill the culture tubes on ice for 10-20 minutes.
8. Transfer the culture aseptically into sterile vials. (5 vials with 0.5ml each)
9. Spin down at 6000 rpm for 8 minutes, preferably in a refrigerated centrifuge at 4°C or spin at Room temperature (RT).
10. Discard the supernatant.
11. Resuspend the cell pellet very gently in small volume of ice-cold solution A (approximately 2 ml), using a pre-chilled pipette. Care must be taken not to remove the tubes from ice during resuspension. Add remaining 33 ml of solution A, resuspend gently.
12. Keep the vials on ice for 5 minutes. Centrifuge at 8000 rpm for 5 minutes at 4°C or spin at RT.
13. Discard the supernatant and chill the tube on ice. Resuspend the pellet in 3 ml of ice-cold solution A.
Note: Resuspension is to be done gently as cells are very fragile at this stage.
14. Heat inactivate the ligated samples at 65°C for 10 minutes. Spin at 5000 rpm for 2 minutes and keep the vials on ice.
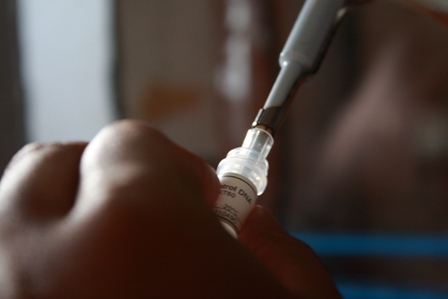
15. Aliquot 2 μl (10 ng) of control DNA each into 5 ligation vials
Transformation:
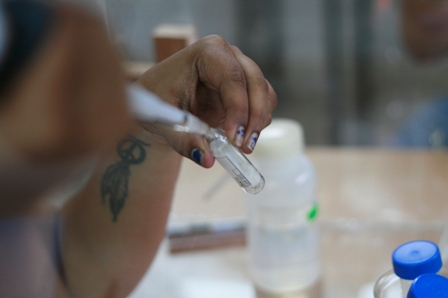
16. Take a 2 vials of ligated sample and control DNA and 1 vial of only control DNA( positive control).
To this add 200 μl each of competent cells. Tap the
vials gently and incubate on ice for 20 minutes.
17. Label the remaining 200 μl of competent cells as non transformed (negative control)
cells, place on ice till the plating step (step
23).
18. Tap all the vials gently and incubate on ice for 20 minutes.
19. Heat shock the cells by placing the vial(s) in 42°C water bath for 2 minutes, then return the vials to ice and chill for 5 minutes.
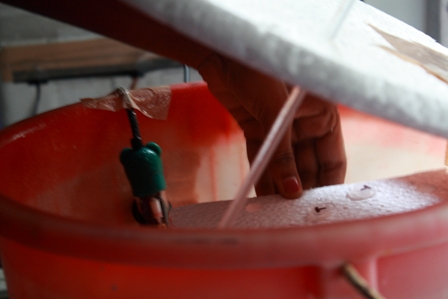
20. Add 0.5 ml of LB broth aseptically to the vial(s) and incubate at 37°C (shaker) for an hour. This is to allow bacteria to recover and express the protein.
Note: While the heat shock treatment is going on, prepare the LB-Amp agar(200 μl in 200 ml of LB agar.)
.(incubate pic)
21. liquify the already made LB-amp agar and pour about 20 to 25 ml of i in each plate.
Note: Wait for agar to solidify again.
22. Pipette 200 μl each from the vials transformed with the ligated mix onto LB-Amp plates and spread thoroughly using spreader/pipette.
23. Pipette 200 μl of LB onto a LB -Amp plate and spread 20 ìl of the cells transformed with control DNA. Label this as positive control plate.
24. Plate 200 μl of non-transformed cells onto another LB-Amp plate to check for contamination. Label this as non-transformed (negative control) plate.
25. Keep one plate with only LB-Amp Agar and nothing else. Name this as double negative control.
26. Incubate the plates at 37°C, overnight.
spreader incubate
Day 3:
Moment of Truth
27. Observe the plates under UV-light (312 nm).
Note: If observed add 254 nm of less intensity of glow could vary.