Explorations in BioLuminescence
From fireflies to stinky squids... the future is bright
Contents
General Introduction
Bioluminescence is visible light made by living creatures. Such creatures are rare on land but extremely common in the oceans. see more on Marine Bioluminescence by BioScience | Explained here: File:BiolumEN.pdf
Bioluminescence is not the same as phosphorescence
Even though Steinbeck, Hemmingway and even Darwin referred to the “phosphorescence of the sea” this is a literary rather than a scientific description. Phosphorescence is the delayed emission of light from a source that has been excited by light. Examples include glow-in-the-dark paints and toys.
Bioluminescence is not the same as fluorescence
As with phosphorescence light emission is stimulated by light not by a chemical reaction. With fluorescence the excitation wavelength is always shorter, that is, higher energy, than the emission wavelength and emission ceases as soon as the excitation source is turned off. Some of this confusion may have arisen because some, but not all, luciferins are fluorescent and a few pass their excitation energy along to fluorescent proteins like GFP.
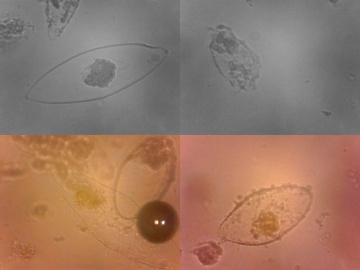
Isolation of luminous bacteria from fish or squid
from bioScience | Explained, download File:BioSci expl PhotoLumen.pdf
P. phosphoreum is one of the commonest spoilage organisms of fish such as cod. It is not known to cause disease, but there are reports of people being startled by glowing fish fingers in the fridge!
To isolate glowing bacteria from fish, obtain a freshly-caught seafish or squid. It is very important that the fish has not been frozen or washed in fresh water. It is also better if the fish has not been stored on ice. Place the fish in a container with 3% NaCl solution. The liquid should be deep enough to come half way up the fish.
Cover the container and store the fish for 24 hours at about 12–15 °C. Note: if this temperature is difficult to achieve, place the fish in a fridge at about 4 °C for 48–72 hours.
After incubation, take the container with the fish into a dark room. When your eyes have been adopted to dark, light spots will be visible on the skin of the fish. Use a sterile toothpick or disposable sterile loop or needle to aseptically transfer the brightest spots onto sterile fish enrichment agar plates. Tip: some people find it useful to use a dim red lamp (e.g., a photographic safety lamp) for this step. Turn the fish away from the lamp so that the glowing colonies are in the shade and therefore visible.
Transfer the cultures to new agar plates every second day if you are incubating them at temperatures around 12 °C or once a week if you are storing them in a fridge. P. phosphoreum will grow at 4 °C; V. fischeri will not. By selecting the brightest colonies when inoculating, it should be possible to isolate a pure culture.
Fish enrichment medium
- Boil 250 g fish meat in 1 l of water.
- Add 30 g NaCl and sieve to remove solids.
- Add 10 g peptone, 10 ml glycerol and 1 g yeast extract.
- Adjust the pH to 7.
- Autoclave at 121 °C for 15–20 minutes.
For a solid medium add 15–20 g of agar to every litre of broth.
Safety note
Several species of Vibrio are pathogenic. The chances of inadvertently isolating pathogens in this procedure can be reduced by using at least 3% salt solution and incubating fish and plates at no more than 15 °C. Human pathogens are unlikely to grow under such conditions.
Further links
GlowShrooms
For various recipes on making agar growth plates check Agar is the Media
A full description of ideal growth conditions has been publisher here http://jresearchbiology.redolences.com/documents/RA0190.pdf
Influence of culture conditions on mycelial growth and luminescence of Panellus stipticus
IB Prasher, VC Chandel, AS Ahluwalia - Journal of research in Biology (2012) 3: 152-159
ABSTRACT:
Panellus stipticus (Bull.) P. Karst, a naturally bioluminescent tropical fungus, has been studied in vitro for the optimum culture conditions viz culture media, temperature, pH and days of incubation required for luminescence. Temperature and pH affect growth and bioluminescence to a great extent. Glucose-peptone medium has been found to be the best for optimum mycelial growth as well as luminescence. The fungus exhibits luminescence at 20-24°C. The maximum mycelia dry weight (mg/25ml of the basal media) and luminescence observed at pH 4.0. The fungus exhibits luminescence after eight days of incubation at 24°C and pH 4.0, whereas it intensified to maximum after 13 days of incubation (pH 4.0 and temperature 24°C).
tips for Shroom Cultures
- bags: sterilizable bags
- Recipes: Agar Medium
Bioluminescent Algae
DINOFLAGELLATES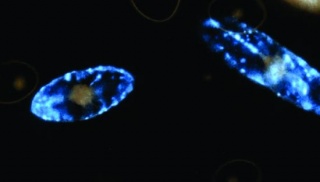
Material for growing dinoflagellates:
A clear growing container (shallow containers with lots of surface area)Sanitation is necessary so your batch doesn‘t crash. REALLY wash out the grow container, make sure there is absolutely no residue left. Culture flasks -- Sterilized glassware if autoclave is available, otherwise use disposable tissue culture flasks. If you use sterile media and glassware, your cultures will continue forever; every month pour about 1/4 of the culture into some new medium. If you can‘t maintain sterile culture conditions, the cells will last for a few weeks to a month before bacteria overgrow the culture.
Water
- Use purified water as tap water can contain chlorine or other things that might kill your batch.
- No tap water, tap water may contain metal ions that are detrimental to algal growth.
- Temperature: 22° C
Sea Salt from pet or aquarium stores
Light and timer
- Indirect natural light is good, but not direct sunlight.
- Illumination with cool-white fluorescent lamps
- Fluorescent light, either cool white or full spectrum, is also sufficient. Avoid incandescent lamps, as they can overheat the water and kill your algae.
- A grow light or an aquarium/terriarium light
- 12 hours of light and 12 hours of darkness every 24 hours
Nutrients (Growth medium)
- Dinoflagellates require nitrate, phosphate, trace metals, and vitamins.
- (Micro Algae Grow)
- Growth medium:
- Dino Food
- Dino Refill
A Starter Culture
- A15 Bioluminescent Algae 250ml cultures from Sciento.
- Planktonic marine, bioluminescent dinoflagellate (Pyrocystis sp.)
Supplier of Dinoflagellates:
- Sciento Manchaster UK http://www.sciento.co.uk/catalog/item/553/
- Empco Holdings Menlo Park USA http://empco.org/edu/index.php/bioluminescent-algae.html
- Sunnyside Sea Farms Goleta California USA http://seafarms.com/html/products.html
- Carolina Burlington USA
Art / Design / Products using dinoflagellates
- Dino Pet Powered by dinoflagellates We like to think of it as lighting in a bottle, only without the pesky thunder.
- Dino Pet at Kickstarter
 from biopop.com
from biopop.com - Sophia Sobers Heartbeats & Bioluminescence / Necklace, heartbeat monitor is a concept
 from Sophia Sobers
from Sophia Sobers
Further links
- Bioluminescent Hourglass using Dinoflagellatets by BioCurious
- Grow Your Own Bioluminescent Algae on Instructables
- Microbial Home Probe project by Philips
Hacking Fireflies
Artists working with the topic of firefly
DIY Bioluminescent Dormice
Luminous Workshopology
interested Brian Degger
- --- the future is bright ---
- Why the hell should everything glow?
Phototaxis is a kind of taxis, or locomotory movement, that occurs when a whole organism moves towards or away from stimulus of light.This is advantageous for phototrophic organisms as they can orient themselves most efficiently to receive light for photosynthesis. (wikipedia.org)
Phototropism is the movement or growth of organisms in response to lights or colors of light (e.g.- the sunflower) (wikipedia.org)
Heliotropism is the motion of plant parts (flowers or leaves) in response to the direction of the sun. A. P. de Candolle called this phenomenon in any plant heliotropism (1832). It was renamed phototropism in 1892, because it is a response to light rather than to the sun. (wikipedia.org)
Towards a mini citizen science festival in Zuri
starting with experimentations
Planning the events
BioLuminescence@Corner's College
other links and reference
Lumière Vivante, Dubois 1900
Classic publication on using bioluminescence to lighting: "Sur l'éclairage par la lumière froide physiologique, dite lumière vivante", Raphael Dubois, Issue J. Phys. Theor. Appl., Volume 9, Number 1, 1900, Page(s) 589 - 591
Leuchtende Pflanzen, Molisch 1904
B. Ueber das Leuchten von Würsten
Ueber diesen Gengenstand finden sich in der Literatur nur wenige brauchbare Nachrichten. Das Beste darüber verdanken wir Heller (1). Er erhielt im Winter 1852 in Wien von dem Sanitätsmagister Dr. Stuhlberger mehrere Würste, welche die Polizeibehörde deshalb in einem Verkaufsgewölbe konfiszieren liess, weil die Würste durch ein sehr starkes Leuchten auf ihrer ganzen Oberfläche verdächtig erschienen. Heller gibt davon folgende Beschreibung: "Die Würste waren rohe, sogenannte "Augsburger", 4 Zoll lang und 1 1/2 Zoll dick. Sie verbreiteten nicht im geringsten einen unangenehmen oder gar fauligen Geruch und hatten einen guten frischen Geschmack. Die Füllung bestand meist aus rohem Rindfleisch und wenig rohem Schweinefleisch.
Die Würste waren weich, an der Oberfläche nass und schleimig und trockneten nicht an der Luft. Sie hatten übrigens am Tageslicht keine besondere Eigenschaft. Schon im Halbdunkel leuchteten sie, aber in einem finsteren Raume war auf ihrer ganzen Oberfläche ein sehr starkes, weisslich-grünliches, ruhiges, nicht dampfendes Licht verbreitet, dass man schon bei einer Wurst gewöhnliche Druckschrift recht gut lesen konnte.
The Mushroom Cultivator, Stamets and Chilton, 1984
Sterile Technique and Agar Culture
Water quality (its pH and mineral content) varies from region to region. If living in an area of questionable water purity, the use of distilled water is advisable. For all practical purposes, however, tap water can be used without harm to the mushroom mycelium. A time may come when balancing pH is important—especially at spore germination or in the culture of exotic species. The pH of media can be altered by adding a drop at a time of 1 molar concentration of hydrochloric acid (HCL) or sodium hydroxide (NaOH). The medium is thoroughly mixed and then measured using a pH meter or pH papers. (One molar HCL has a pH of 0; one molar NaOH has a pH of 12; and distilled water has a pH of 7).
After thoroughly mixing these ingredients, sterilize the medium in a pressure cooker for 30 miutes at 15 psi (equals 103 KPa). (Pressure cookers are a safe and effective means of sterilizing media provided they are operated according to the manufacturer's instructions). A small mouthed vessel is recommended for holding the agar media. If not using a flask specifically manufactured for pouring media, any narrow necked glass bottle will suffice. Be sure to plug its opening with cotton and cover with aluminum foil before inserting into the pressure cooker. The media container should be filled only to 2/3 to 3/4 of its capacity.
Place the media filled container into the pressure cooker along with an adequate amount o water for generating steam. (Usually a 1/2 inch layer of water at the bottom will do). Seal the cooker according to the manufacturer's directions. Place the pressure cooker on a burner and heat until ample steam is being generated. Allow the steam to vent for 4-5 minutes before closing the stop cock. Slowly bring the pressure up to 15 psi and maintain for 1/2 hour. Do not let the temperature of the cooker exceed 250 °F. or else the sugar in the media will caramelize. Media with caramelized sugar inhibits mycelial growth and promotes genetic mutations. A sterilized pot holder or newly laundered cloth should be handy in the sterile lab to aid in removing the media flask from the pressure cooker. While the media is being sterilized, immaculately clean the laboratory.
http://www.amazon.com/The-Mushroom-Cultivator-Practical-Mushrooms/dp/0961079800
Nice Poster
Poster designed by Seattle-based artist Eleanor Lutz