Nata de Coco
Contents
- 1 Cellulose producing Bacteria
- 2 Cultures and applications
- 3 New Experiments in Lifepatch 2022
- 4 Isolating ナタ菌 Komagataeibacter xylinus (formerly known as Nata de Coco ナタ・デ・ココ) - Oki WonderLab 2020
- 5 Experiments in Lifepatch 2019
- 6 Experiments at Home - Zürich
- 7 How to make it
- 8 Where to get it?
- 9 Further info
Cellulose producing Bacteria
Food and edible jargon
It seems a much more common thing than one thinks... Easy to find on rotten fruits, in beer or wines, simple to isolate and ready to use for edible products etc. Kombucha brewers welcome (File:2019_kombucha_bookChapter.pdf)
While kombucha is a complex symbiotic mixture of different organisms, other pure cultures can be isolated of cellulose producing bacteria, usually in the family of Acetobacter xylinum or related, now taxonomically called Komagataeibacter xylinus.
While many scientists talk about "novel nano materials produced by bacteria", BNC (Bacterial NanoCellulose), seemingly invented in the 90ies... it turns out under the name Nato de Coco to be a commercial edible desert product for a loooong time coming from the Phillipines and also very wide-spread in Indonesia. Easy to get the started cultures from the local tradional markets or agro supply stores. See below.
http://www.ricebowlasia.com/questions/what-is-the-manufacturing-process-of-nata-de-coco/
"Homemade Nata de Coco (super easy with only 3 ingredients)"
Nata de coco is common in Indonesian desserts, and probably also in some Asian desserts. We all love nata de coco, especially when it is added to pudding mixture or sweet drinks. It is so refreshing and delicious.
Lately, I found some very easy recipes to make nata de coco at home. This was so easy and I didn’t even need fresh coconut flesh. All I needed were jelly powder, coconut water, and sweetened condensed milk. Yes, only 3 ingredients and it only took less than 5 minutes to make it. You can add sugar if you want it to be sweeter.
This was good and absolutely healthier than store bought nata de coco. I still keep them in the fridge (and I can’t stop munching on them) and hopefully, I can make some sweet dessert before I finish them all by myself……….
Sciency talk
On an acetic ferment which forms cellulose
Brown, J. Chem. Soc., Trans., 1886,49, 432-439 https://doi.org/10.1039/CT8864900432 Download book here: File:Bacterial Nanocellulose.pdf
DURING my work on the chemical actions of Bacterium aceti, described in a previous paper (this vol., p. 172), I met with the peculiar acetic ferment commonly known as the “vinegar plant” or “mother”. This ferment differed so much in appearance from any form of B. aceti I had noticed, that it seemed probable it was a distinct organism. In order to ascertain this, and also to enable me to study its chemical actions, I obtained pure cultivations by a combination of the fractional and dilution methods in the way I described in my previous paper. The nutrient solutions used for this purpose were composed of red wine diluted with half its bulk of water, and rendered acid with 1 per cent. of acetic acid in the form of ordinary vinegar. This liquid strongly favours the growth of the acetic ferments, and is at the same time very prejudicial to the growth of most other organisms.”
"Nanocelluloses: A New Family of Nature‐Based Materials"
Klemm, D. , et. al (2011), Angew. Chem. Int. Ed., 50: 5438-5466. doi:10.1002/anie.201001273 Download .pdf here: File:2011_Nanocelluloses.pdf
When BNC fleeces were submerged in a dispersion of multiwalled carbon nanotubes (MWCNTs) for several hours, individual MWCNTs adhered strongly to the surface and inside of the BNC fleeces. Conductivity measurements demonstrated that the incorporation of carbon nanotubes is a suitable way to prepare electrically conductive BNC membranes.
Transparent and electrically conducting films were also fabricated by the adsorption of single-walled carbon nano-tubes on bacterial cellulose membranes embedded in a transparent polymer resin. In this way, films with a wide range of transmittance and surface-resistance properties could be obtained by controlling the immersion time and carbon-nanotube concentration. A transparent conducting film with a transmittance and surface resistance of 77.1 % at 550 nm and 2.8 kW sq �1 , respectively, was fabricated from a 0.01 wt % carbon-nanotube dispersion during an immersion time of 3 h. The transparent conducting films were quite flexible and maintained their properties even after crumpling.
BNC layers have also been investigated as loudspeaker vibration films. It was demonstrated that these films have the advantages of simple manufacturing by bacterial biofabrication, good mechanical properties and thermal stability, good fundamental characteristics of a sound-vibration film, high specific elasticity and loss factor, long service life, and environmental friendliness.
Highly flexible, transparent, and conductive silver nanowire-attached bacterial cellulose conductors
Lv, P., Zhou, H., Zhao, M. et al. Cellulose (2018) 25: 3189. https://doi.org/10.1007/s10570-018-1773-8 download .pdf here File:2018_HighlyFlexibleTransparentAndCo.pdf
 link title
A simple, rapid method for developing conductive, ultrafine, and high aspect ratio silver nanowires (AgNWs) is reported. Transparent and flexible nanocomposites were fashioned from bacterial cellulose (BC) and AgNWs in a very straightforward and direct manner without the addition of materials or the need for specific facilities. The as-prepared BC/AgNWs composite thin films were able to demonstrate acceptable transparency (near 80% at 550 nm), high flexibility, good mechanical strength (18.95 MPa) and stable conductivity (7.46 Ω sq−1) under various bending states.
link title
A simple, rapid method for developing conductive, ultrafine, and high aspect ratio silver nanowires (AgNWs) is reported. Transparent and flexible nanocomposites were fashioned from bacterial cellulose (BC) and AgNWs in a very straightforward and direct manner without the addition of materials or the need for specific facilities. The as-prepared BC/AgNWs composite thin films were able to demonstrate acceptable transparency (near 80% at 550 nm), high flexibility, good mechanical strength (18.95 MPa) and stable conductivity (7.46 Ω sq−1) under various bending states.
Bacterial NanoCellulose - A Sophisticated Multifunctional Material
Miguel Gama, Paul Gatenholm, Dieter Klemm, 2017, CRC Press
Bacterial nanocellulose (BNC) is an emerging nanomaterial with unique properties produced by several species of ubiquitous fermentation bacteria, most importantly Gluconacetobacter xylinus, previously known as Acetobacter xylinum. BNC has been used for a variety of commercial applications including textiles, cosmetics, and food products, and it has a high potential for medical applications.
Bacterial NanoCellulose: A Sophisticated Multifunctional Material provides the state of the art of scientific knowledge about the mechanism of cellulose production by bacteria along with pointing out challenges in expansion of BNC production in large scale. It provides the latest update on BNC structure and its modification, as well as comprehensive information about current and future applications of BNC.
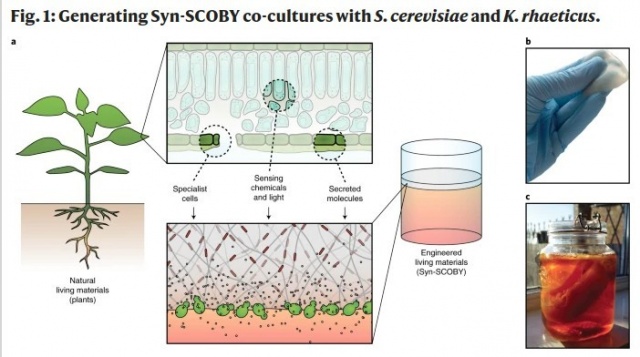
Living materials with programmable functionalities grown from engineered microbial co-cultures
Biological systems assemble living materials that are autonomously patterned, can self-repair and can sense and respond to their environment. The field of engineered living materials aims to create novel materials with properties similar to those of natural biomaterials using genetically engineered organisms. Here, we describe an approach to fabricating functional bacterial cellulose-based living materials using a stable co-culture of Saccharomyces cerevisiae yeast and bacterial cellulose-producing Komagataeibacter rhaeticus bacteria. Yeast strains can be engineered to secrete enzymes into bacterial cellulose, generating autonomously grown catalytic materials and enabling DNA-encoded modification of bacterial cellulose bulk properties. Alternatively, engineered yeast can be incorporated within the growing cellulose matrix, creating living materials that can sense and respond to chemical and optical stimuli. This symbiotic culture of bacteria and yeast is a flexible platform for the production of bacterial cellulose-based engineered living materials with potential applications in biosensing and biocatalysis.
Full paper for download here: File:2020_LivingMaterial_synSCOBY.pdf
More references:
- 2012 - Altering the growth conditions of Gluconacetobacter xylinus to maximize the yield of bacterial cellulose
- 2019 - Bacterial nanocellulose: Present status, biomedical applications and future perspectives
- 2018 - Bacterial cellulose yield increased over 500% by supplementation of medium with vegetable oil
- Nanocellulose - From Nature to High Performance Tailored Materials, 2012, Dufresne, Alain
Design and Fashion Style
Successful realization of natural self-grown fashion (SGF) has tremendous creative and practical potential, as well as a profound ecological effect on the fashion industry and the environment. From this publication.
"Growing materials has become more and more popular in design, so maybe growing things is closer and easier than we think," said Janusz.
See this article.
Found a nice company producing reinforced materials from nata de coco "Made from Malai".
Business Jargon
From JeNaCell Company in Germany
epi nouvelle+ naturelle gently supports regeneration of sensitive skin -100% vegan. Free from other ingredients, epi nouvelle+ naturelle completely relies on the cooling, skin-relaxing effect of pure water.
epi nouvelle+ naturelle is therefore very well suited for post-treatment of peels, mesotherapy, laser therapy, skin intolerances such as perioral/ periorbital dermatitis, photodynamic therapy and follow-up treatment in aesthetic or surgical operations.
With the biocellic + product line, we offer a basic material for a variety of innovative technical solutions. The material biocellic+ has a multitude of extraordinary properties: in particular the high mechanical properties and thermal stability and the high chemical resistance.
biocellic + is therefore very suitable for use as a starting material for membranes, as filter materials for separation techniques, as a carrier material for catalysts or as a composite material for the fiber reinforcement of (bio) polymers. It is available as a nonwoven, film, spherical particle or fiber- 100% vegan.
Cultures and applications
Nata de Coco aka Komagataeibacter xylinus
The best!!!
This one is the main bacteria that is commonly used, but has many names and strains....
Komagataeibacter xylinus, formerly known as Acetobacter xylinum or Gluconacetobacter xylinus
Nata de Soya
It's what the honfies used for their SOYA C(O)U(L)TURE
Gluconacetobacter hansenii (present from Isaac from Chile)
What is it?
the name of your traveling companion is Gluconacetobacter hansenii (cellulose strain).
Below is the media for optimal cellulose production, but we use it for culturing etc.
Hestrin–Schramm (HS) medium (for 500mL)
- 2.5g yeast extract (0.5% w/v)
- 2.5g peptone (0.5% w/v)
- 1.35g Na2HPO4 (0.27% w/v)
- 0.75g citric acid (0.15% w/v)
- 10g glucose (2%w/v) - NB autoclave this separately as 20% glucose, and add 50ml to 450ml of the rest of the media
Hestrin–Schramm (HS) medium (for 1L)
- 5g yeast extract (0.5% w/v)
- 5g peptone (0.5% w/v)
- 2.7g Na2HPO4 (0.27% w/v)
- 1.5g citric acid (0.15% w/v)
- 20g glucose (2%w/v) - NB autoclave this separately as 20% glucose, and add 50ml to 450ml of the rest of the media
other tips
I think is no problem with split the amount I gave you. To accomplish it, you should inoculate the media with a piece of the cellulose-bacteria layer and left it growing without agitation at room temperature (you only have to be careful with the contamination at the moment of inoculation!).
You can get a lot more information from this group of iGEM: http://2014.igem.org/Team:Imperial
If you are interested on dye the cellulose with bacterial pigments, we usually do this way:
- grow the cellulose paper
- remove the excess of media
- use the cellulose paper as substrate, by plating (with L-shape spreader) a pigment producer O.N. bacterial culture over the wet cellulose paper.
- Bacterias will grow on that side of the paper and produce the dye. If you want to have both sides dyed, repeat the process on the other side.
I hope it is clear :)
have a good time on Japan and don't hesitate to ask me if you need something.
New Experiments in Lifepatch 2022
Let's find out where to get that Pink Coconut!!
So nice and easy to buy the starter culture here: https://www.tokopedia.com/agrotekno/acetobacter-xylinum-1000-ml
First Batches
How to open a coconut
First batch is growing...
We made 2 recipes: one with the coconut water from the old coconuts, donated by Geger's mom, and one with the juice of 2 fresh young coconuts "kelapa muda". both lead to approx 2 liters of medium.
We kinda added the other ingredients, according to the book: ZA (ammonium sulfate), garam ingris (magnesium sulfate), sugar (garam), citric acid (cos we didnt have normal table vinegar), and dried yeast powder.
It might be we made some mistakes late night mixing the ingredients...
The stuff was then boiled for 20 minutes... and poured into plastic trays.
Next morning when cooled down, the starter was added, 300 ml each or something. but where did we add the rest?
After one week... we checking the growth. From the young Coconut media just some slime on the bottom. no growth. The other one showed a thin layer, but kinda bubbly surface structure on the lower side. And dusjagr couldn't wait, and took it out to play with it!
Pressing and drying the first batch
Boiling and soaking in NaOH
Second and Third batch
Still working with the coconuts and finally found the PINK ones!!!
Ordered new starters from another company. They also sell small tubes for 1l each. See more Nata_de_Coco#How to make Nata De Coco practically and easily with a small tube starter:. This could be useful to ship to friends and bring home. Estimate i can have them outside for 1-2 weeks, or in the fridge for 1-2 months.
Starter kit for Take-san
Testing different growth medias
Using labstyle compositions, higher sugar content could lead to faster growth and higher yield.
From this publication:
- 2012 - Altering the growth conditions of Gluconacetobacter xylinus to maximize the yield of bacterial cellulose Download here: File:Altering the growth conditions of Gluconacetobacter xylinus_Ruka_2012.pdf
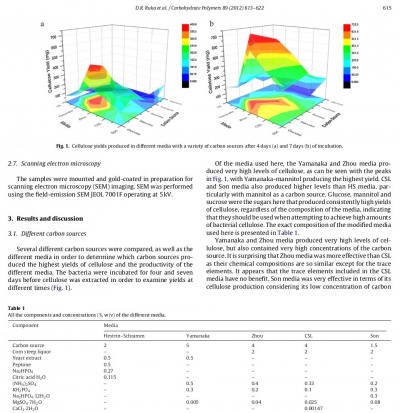
The exact composition of the media is described below. All media were adjusted to pH 5.0 with HCl or NaOH and autoclaved at 121 ◦ C for 20 min. The carbon sources glucose, mannitol, sucrose, fructose and glycerol were substituted in these media.
Seed cultures were prepared by selecting a single colony from a working plate of Hestrin–Schramm agar and inoculating 10 mL of HS broth. These cultures were incubated for seven days at 28 ◦ C under static conditions. Following growth, seed cultures were shaken vigorously to remove the bacterial cells from the cellulose pellicle. Pellicles were removed and the resulting cell suspension was used for inoculations. Cultures were grown in 200 mL conical flasks containing 50 mL of media and were inoculated at a concentration of 1% (v/v) of the cell suspension unless otherwise stated. Cultures were incubated for seven days at 28 ◦ C under static conditions unless otherwise stated.
When investigating surface area, beakers of different sizes were used in order to achieve different surface areas. It was found that 50 mL of media in large beakers often dried out or only produced a very thin layer of cellulose as there was not enough depth for the cellulose to move into the media when the media was spread so thinly. To counteract this, volumes of 100 mL and 200 mL of media were also used in the different sized beakers. The 100 mL and 200 mL cultures were inoculated with 0.5% and 0.25% (v/v) inoculum volumes in addition to the typical 1% (v/v) inoculum, respectively.
Adding vegetable oil, some report says 500% increased yield!!!
- 2018 - Bacterial cellulose yield increased over 500% by supplementation of medium with vegetable oil Download here: File:Bacterial cellulose yield increased over 500%_Zywicka_2018.pdf
Making the conductive Nata
From the publication "Highly flexible, transparent, and conductive silver nanowire-attached bacterial cellulose conductors"
Highly flexible, transparent, and conductive silver nanowire-attached bacterial cellulose conductors
Lv, P., Zhou, H., Zhao, M. et al. Cellulose (2018) 25: 3189. https://doi.org/10.1007/s10570-018-1773-8 download .pdf here File:2018_HighlyFlexibleTransparentAndCo.pdf
A simple, rapid method for developing conductive, ultrafine, and high aspect ratio silver nanowires (AgNWs) is reported. Transparent and flexible nanocomposites were fashioned from bacterial cellulose (BC) and AgNWs in a very straightforward and direct manner without the addition of materials or the need for specific facilities. The as-prepared BC/AgNWs composite thin films were able to demonstrate acceptable transparency (near 80% at 550 nm), high flexibility, good mechanical strength (18.95 MPa) and stable conductivity (7.46 Ω sq−1) under various bending states.
let's see how "simple" or "straightforward" and "no need for specific facilities" this really is...
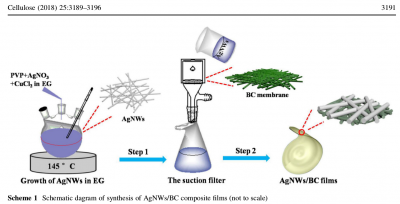
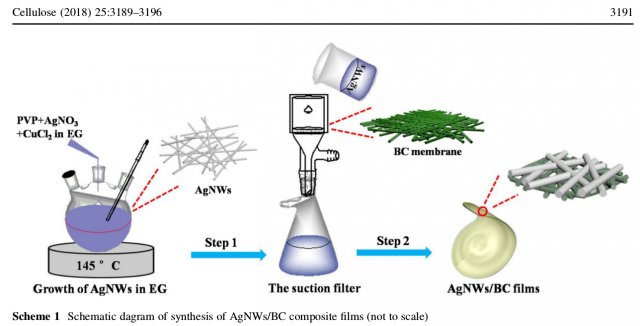
The details of the synthesis procedure are described as follows:
BC pellicles were produced by cultivating K. Xylinus in the classical Hestrin and Schramm (HS) medium that contained 5 g L -1 yeast, 3 g L -1 bacto-peptone and 25 g L -1 D -mannitol. The flasks were incubated at 30 °C for 4 days in a static incubator. The as-prepared cellulose was dipped into 1 wt% NaOH at 80 °C to remove remaining bacterial and culture liquid, and subsequently washed several times until neutral pH.
The AgNWs synthesis procedure was as follows:
100 mL EG was heated at 170 °C for 1 h in oil-bath pan to remove water. * 0.2 g PVP as stabilizer, 0.17 g AgNO 3 , and 2.4 mg CuCl 2 were all dispersed into 60 ml EG in hotplate by stirring for 30 min to ensure uniform dispersion. The mixture was added into a 100 ml beaker and heated at 145 °C for 6 h in oil-bath pan. The precipitate was centrifuged three times under 1000 rpm for 3 min with acetone and centrifuged three times to obtain AgNWs in ethyl alcohol. Lastly, under the condition of optimum ratio, 10.8 mg AgNWs was dispersed into 200 mL ethyl alcohol and filtered on the as-prepared single-sided BC matrix, as shown in Scheme 1.
Another way to make conductive transparent layer with guava seeds and RISO PRINT GOCCCO LAMPS
Stuff to prepare
Isolating ナタ菌 Komagataeibacter xylinus (formerly known as Nata de Coco ナタ・デ・ココ) - Oki WonderLab 2020
Mostly documented and discussed on the hackteria forum:
https://forum.hackteria.org/t/isolating-komagataeibacter-xylinus-formerly-known-as-nata-de-coco/836
Experiments in Lifepatch 2019
Experiments at Home - Zürich
Recipes
The "pure" medium
Kinda like the HS medium...
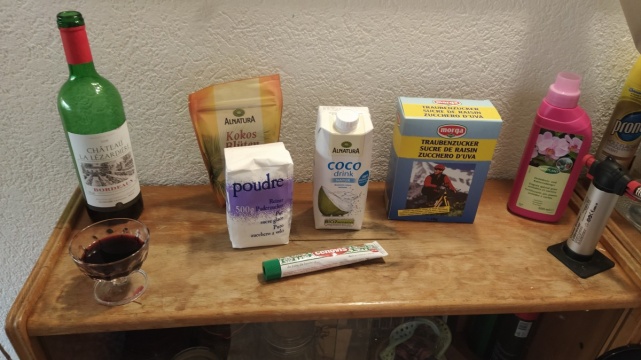
- First the glucose ("Traubenzucker" also known as dextrose) was boiled for 15 min, 40g / 200 ml "destillatgleiches Wasser" (kinda distilled water).
- 0.8 l of "destillatgleiches Wasser" and a little bit of tap-water, boiled for a while
then adding:
- 5g Yeast Extract (really old one i found in my box)
- 5g Peptone
- 1.5g Citric Acid
- 2.7g K2HPO4 (Monopotassium phosphate or Dipotassium phosphate? I forgot... had only Potassium instead of Sodium...)
- kinda 50 ml of the glucose solution... maybe 13-18g of glucose
- 0.08 l of Acetobacter xylenum starter-culture
Swiss Kitchen Recpie
- 3l Water
- 20g Cenovis
- 4.5g Citric Acid
- 3g Fertilizer, liquid (NPK 4-5-6 with trace elements, Mioplant Orchideendünger)
- ca 25g of Glucose aka "Traubenzucker"
Boiled for 10 min in a pot, then left to cool down in 1.5 l glassjar, adding few small pieces of pellicle from starter culture, and flat plastic bowl. After cooling adding the following
- Jar: 0.08 l of Acetobacter xylenum starter-culture
- Bowl: 0.15 l of Acetobacter xylenum starter-culture
Coconut Recipe
- 0.7l Coconut water (Alnatura Coco drink)
- 2l Water
- 4.5g Citric Acid
- 3g Fertilizer, liquid (NPK 4-5-6 with trace elements, Mioplant Orchideendünger)
- 1g K2HPO4 (Dipotassium phosphate)
- 3 tablespoons of Glucose aka "Traubenzucker"
Boiled for 10 min in a pot, then left to cool down in 1 l glassjar, adding few small pieces of pellicle from starter culture, and flat plastic bowl. After cooling adding the following
- Jar: 0.08 l of Acetobacter xylenum starter-culture
- Bowl: 0.1 l of Acetobacter xylenum starter-culture
Akbar's Super Fermentation Recipe
How to make it
DIBTB - Do it by the book!
Download the translated book here: File:Nata_de_Coco-Book_translated_small.pdf
And just found this tool to convert pages of a book into a foldabe booklet with ubuntu.
pdfbook2 ./Pin_BioTransLab.pdf --short-edge --resolution=300 --outer-margin=0 --inner-margin=30 --top-margin=0 --bottom-margin=0
NATA DE COCO FROM COCONUT WATER
Taken from this website
Materials:
- Coconut water
- Acetic acid
- Refined sugar
- Nata starter
- Ammonium phosphate
Equipments:
- Weighing scale
- Strainers
- Wide-mouthed glass jars or basins
- Kettles
Procedure:
- The collected coconut water is filtered through a cheesecloth. One hundred (100 gms.) refined sugar and 5 grams monobasic ammonium phosphate is mixed for every liter of coconut water in a container. The container is covered and the mixture allowed to boil. It is then allowed to cool after boiling and 6.9 ml. of glacial acetic acid is added.
- 110-150 ml. of starter (available at ITDI) is added to the mixture. It is subsequently transferred to big mouthed clean jars leaving ample space atop mixture and covered with clean cheese cloth. The culture is allowed to grow at room temperature for 15 days or more. Note: Do not move jars during growth period.
- Harvest is ready after 15 days or more, making sure that all conditions are aseptic so as to enable one to reuse the remaining liquid which serves as starter for succeeding preparations.
Dessert Making. The “nata” is cut into cubes and is subjected to a series of boiling with fresh water until acidity is totally removed. One kilo of refined sugar is added for every kilo of nata and are mixed. It is brought to boiling until the “nata” cubes become transparent.
How to make Nata De Coco practically and easily with a small tube starter:
1.) 1 liter of coconut water is filtered and then put in a panic and cooked until it boils
2.) When it boils, add 25 gr white sugar (2 spoons), 5 gr ZA (1 tea spoon), 10 ml vinegar (2 spoons) - you can use a different ratio scale - then mix well. Furthermore, we call this coconut water stew as coconut water dough.
3.) If foam appears on the surface of the coconut water, the foam must be removed.
4.) Pour the coconut water mixture (while still hot) into the prepared plastic tray and then cover it tightly with paper in such a way that it becomes a nursery tray. You can use rubber or duct tape.
5.) Wait for the coconut water mixture to be cold (approximately 30 C or equal to room temperature)
6.) Open the edge of the paper cover of the tray containing the boiled coconut water that has cooled, then pour the small tube of nata starter. Pour directly without intermediaries and without stirring.
7.) Cover tightly so that no dust, dirt or small animals enter. Store trays properly and neatly. The tray must not be disturbed or shaken during the fermentation process
8.) Let the nursery tray soak 7 to 10 days
9.) The thicker the coconut water mixture in the tray, the longer the fermentation process and vice versa
10.) Open the seedling yield tray after sufficient time. Newly harvested nata sheets are usually slimy, have a sour and sometimes unpleasant aroma
How to harvest nata de coco
- Nata will be in sheet / flat form. Take and remove away the slime. Cut off the damaged or fungus affected parts. Then soak and clean the thin layer (epidermis) if there is any sticking to the surface. Soak again with clean water for 1 day (change the water if it is dirty or smells acid / sour. The first day can be up to 3 times of water changing and can be adjusted)
- The second day, replace the soaking water with clean water, then clean the nata sheet again. Cut the nata into cubes (size according to taste). Soak again for 1 day (the procedure can be repeated the same as number 1) to get rid of the sour / acidity smell, the nata soaking water can be added with pieces of pandan leaves.
- On the third day, the nata in the form of cubes is washed again. If it is sufficient enough (the sour smell is gone), the nata is boiled until it boils and the first boiled water is discarded.
- It is again need to boil the nata and mixed with sugar or syrup according to taste.
- Serve and let the nata stew to cool completely before consuming. It's better to add ice to make it feel chewy. Nata de coco is ready to be served.
Small tube version nata starter:
- Can be stored for 3 weeks from delivery
- Can be only for single use in one pour
- Possible to make 3 liters of coconut water in 1 tray
- Storage should be at room temperature
- The starter fluid in the 10 ml tube is not full due to the needy for oxygen (aerobic bacteria)
Where to get it?
Local sources "Nata de Coco"
In Yogyakarta
https://www.tokopedia.com/search?st=product&q=nata%20de%20coco%20starter
AgroTekno Lab Store in Yogyakarta: https://goo.gl/maps/SS2MAxJJTrjnRZKx7
Buy it online
In Japan / Okinawa
https://www.jcm.riken.jp/cgi-bin/jcm/jcm_keyword?AN=Komagataeibacter&BN=xylinus&CN=&DN=
https://www.nite.go.jp/nbrc/catalogue/NBRCCatalogueDetailServlet?ID=NBRC&CAT=00015237
Wild type isolation - Akbar's Workshop Protocal Translated
A. Making nata seedlings
- . Pineapple peeled, washed to produce pineapple meat. Pineapple meat is cut into small pieces, crushed, and the pulp is taken.
- . Pineapple pulp mixed with sugar and cooking water in a ratio of 6: 1: 3 until evenly distributed.
- . Put the mixture in a clean jar and cover in a filter cloth. Let stand for 2-3 weeks.
- . Leave uninterrupted for 2-3 weeks until a white layer or acetobacter xylinum seedlings are formed.
B. How to make nata:
- . Filter coconut water using a filter cloth then bring to a boil and chill.
- . Mix granulated sugar (100 g / l coconut water), 20 ml vinegar acid / l coconut water and Acetobacter xylinum seeds (170 ml) into coconut water in a mixing pan, then stir until evenly distributed. The mixture has acidity (pH) 3-4.
- . Enter the mixture into a natural jar with a mixture height of 4-5 cm, then cover with a cloth. Place the jar in a clean and safe place.
- . After 15-20 days of the fermentation process, a layer of nata is formed on the surface of the liquid with a thickness of 1-2 cm. Nata layer with a weight of + 200 g. The liquid under the nata is the liquid of the seed that can be used for making the next nata.
Harvesting nata.
- . The nata layer is carefully removed using a clean fork or clamp so that the liquid under the layer is not polluted. The liquid under the nata can be used as seedling liquid in subsequent processing.
- . Remove the membrane attached to the bottom of the nata, washed and cut in the form of cubes with a size of 1.5 x 1.5 x 1.5 cm and washed. Pour and soak the nata de coco pieces in a plastic bucket for 3 days and replace the immersion water every day. After that, nata boiled until boiling at 110oC for.
- . For 10-20 minutes. The purpose of soaking and boiling is to eliminate the sour taste.
- . Nata is put in the syrup then simmer at a temperature of 100oC + 15 minutes, after that if necessary can be added to the ingredients of vanilla or other fragrance and salt to taste, then left for 1 night. Make nata syrup in the ratio of 3 kg of cut nata products, 2 kg of sugar and 4.5 l of water are needed. First the sugar is poured into water, heat until dissolved and then strain.
- . Next, nata is packed in a plastic bag or jam jar with a ratio between solids and 3: 1 liquid, the bottle is tightly closed, then boiled in boiling water for 30 minutes. Lift and cool in the air with the lid on the bottom, then the bottle is labeled and ready to be marketed.
Isolating Komagataeibacter xylinus from Pineapple, Oki Wonder Style
Coming soon...
Video instruction
Indo Kitchen Style
Getting the wild type starters from pineapple
Growing your own Nata de Coco at home
More
https://www.youtube.com/watch?v=9EAG2qKW7VM
Same Same but different
Swiss sciency style
Indo Coconut style
Indo Factory style
Philippino 90ies style
Further info
- Highly conductive and stretchable conductors fabricated from bacterial cellulose https://www.nature.com/articles/am201234
- 3D printing with cellulose materials https://link.springer.com/article/10.1007/s10570-018-1888-y
- Biomimetic 4D printing https://www.nature.com/articles/nmat4544#abstract
- Preparation of Eco-Friendly Transparent Electrode for Flexible Device Using Cellulose Substrate https://link.springer.com/article/10.1007%2Fs12221-019-1038-7
- Highly flexible, transparent, and conductive silver nanowire-attached bacterial cellulose conductors https://link.springer.com/article/10.1007/s10570-018-1773-8
- Microbial Production of Biopolymers and Polymer Precursors: Applications and Perspectives https://books.google.co.id/books?id=Vu9kc0-uSJYC
- Aqualose IGEM2014 bacterial cellulose by engineering Gluconacetobacter xylinus http://2014.igem.org/Team:Imperial/Protocols#gluconacetobacter
- How to Make Nata De Coco at Home https://steemit.com/coconut/@the3brother/how-to-make-nata-de-coco-at-home-fresh-menu-and-favors-to-open-fasting-69a0dcd7f935a
- Sequence-based analysis of the bacterial and fungal compositions of multiple kombucha https://www.sciencedirect.com/science/article/pii/S0740002013001846 Download here: File:Sequence-based analysis of the bacterial and fungal compositions ofmultiple kombucha.pdf
- ETHZ Bacterial nanocellulose foams made in 2019: https://www.youtube.com/watch?v=xAZtaAyUhxM
- multi aplikasi: https://www.kompasiana.com/ajuskoto/550e4d4fa33311c12dba80da/multiaplikasi-nata-de-coco